Research Overview
|
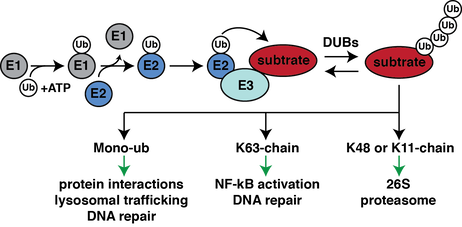
Ubiquitination is a dynamic post-translational modification that regulates biological processes such as cell cycle regulation, protein degradation, metabolism, and immunity. Ubiquitin-activating (E1) enzymes, ubiquitin-conjugating (E2) enzymes, E3 ubiquitin ligases (E3), and deubiquitinating enzymes (DUBs) work in concert with a network of regulatory proteins to establish the cellular ubiquitin landscape, and dysregulation of this landscape contributes to the development of many human diseases including cancer and neurodegenerative diseases. Aberrant ubiquitination may occur as the result of acquired mutations in host genes or through the action of genes encoded by pathogens. Gaining a deeper understanding of human disease states linked to dysregulation of ubiquitin pathways will shed light on how these diseases develop, which in turn may lead to new methods of diagnosis and therapy.
|
The ubiquitin cascade is hierarchically organized with two E1, ~40 E2, and >600 E3 proteins. This highly orchestrated enzymatic process results in diverse ubiquitination profiles that can influence many different cellular processes.
|
Given the importance of the ubiquitin system to cellular survival, it is not surprising that pathogens hijack the ubiquitin machinery during various stages of their life cycles. By studying how oncogenic viruses re-wire critical ubiquitin circuits, we will gain valuable insight into both viral pathogenesis and human diseases linked to aberrant ubiquitination events.
|
HPV hijacks cellular ubiquitin complexes
|
Human Papilloma Virus (HPV) is the most common sexually transmitted disease worldwide and is responsible for nearly all cervical and anal cancers, and for the majority of vaginal, vulvar, penile, and oropharyngeal cancers. Of the >100 different types of HPV, only a subset (high-risk HPVs) are associated with cancer. We are interested in HPV as it provides an exciting opportunity to study the role of ubiquitination in cancer. Nearly half of the viral proteins encoded by HPV, including well-established oncoproteins, can interact with cellular E3 ubiquitin ligases. We aim to define the molecular mechanisms employed by HPV oncoproteins to reprogram the function of host E3 ligases. Our research incorporates techniques in protein biochemistry, biophysics, structural biology, and virology. We envision that a deeper understanding of intermolecular interactions between oncoproteins and host ubiquitin machinery will uncover new regulatory mechanisms related to the broader role of ubiquitination in cancer.
We are also interested in developing therapeutic modulators of ubiquitin E3 ligases. The overwhelming complexity of the ubiquitination system coupled with the presence of difficult-to-target protein-proteins interfaces (PPIs) has created a major barrier to the development of drugs that interfere with ubiquitin transfer. Therapeutic targeting of E3 ligases will, therefore, require strategies that can identify new “molecular vulnerabilities” in the structures of E3 ligases that may be exploited for therapeutic intervention. In addition to our structural studies, we will generate biologics against viral E3 ligases to serve as biochemical probes to reveal structure-function relationships and to expose novel targets for small molecule drugs.